Why Abzena?
Trust our focused approach.
To balance the scales more favorably and mitigate some of the biggest risks in drug development, it is essential to gather as much information about a drug candidate early on to make better, more informed decisions further down the line. This is where the idea of ‘developability’ comes in: developability refers to the assessment of a drug candidate’s properties and potential risks to determine its suitability for further development. Developability helps balance the scales of an ideal drug candidate vs the challenges of reality.
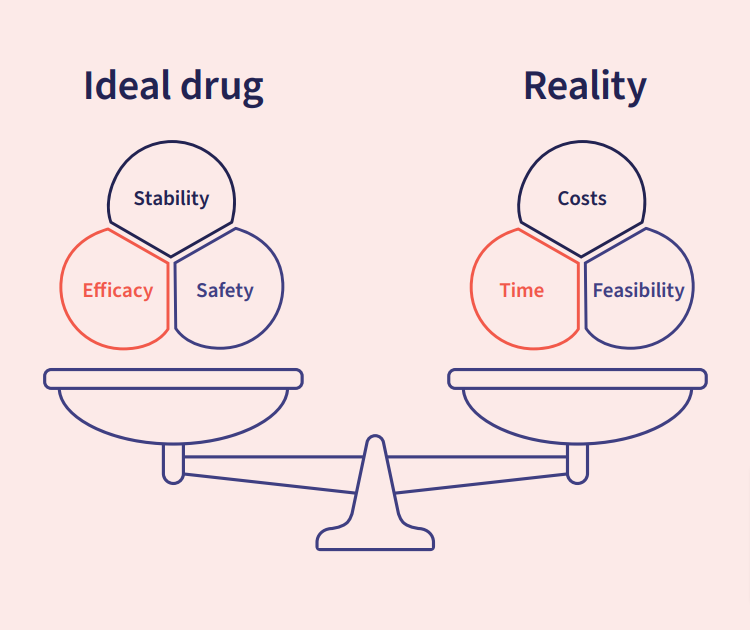
Designing a drug means balancing all the desirable features of a perfect candidate with the reality of pressures faced during discovery and development.
Reality: Costs, Time, Feasibility
To add context, it’s estimated that for each new drug to go from idea to being approved for clinical use, it takes over 10–15 years with an average cost of over $1–2 billion.
Average cost
Average time
Rate of failure
Source: (Sun D et al. (2022) Acta Pharm Sin B. Jul;12(7):3049-3062)
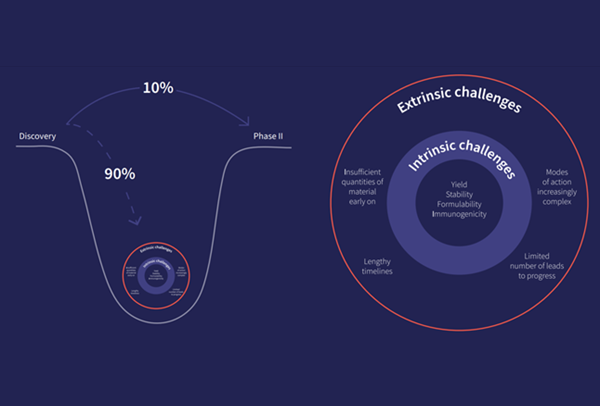
Common reasons for attrition
Impact of attrition
How do Abzena allow you to start smart and finish fast? Read more below or contact our team of experts today to learn more about our developability assessment approach.
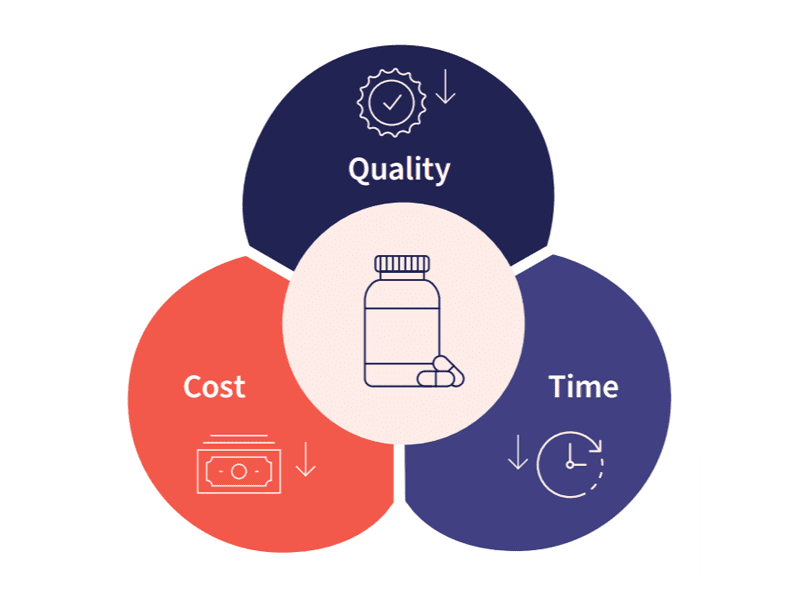
Our approach to Developability Assessment helps to mitigate risks by providing a thorough understanding of your molecules.
Developability Assessment aims to:
At Abzena, we understand that assessing developability represents a strategic solution that guides the decision-making process.
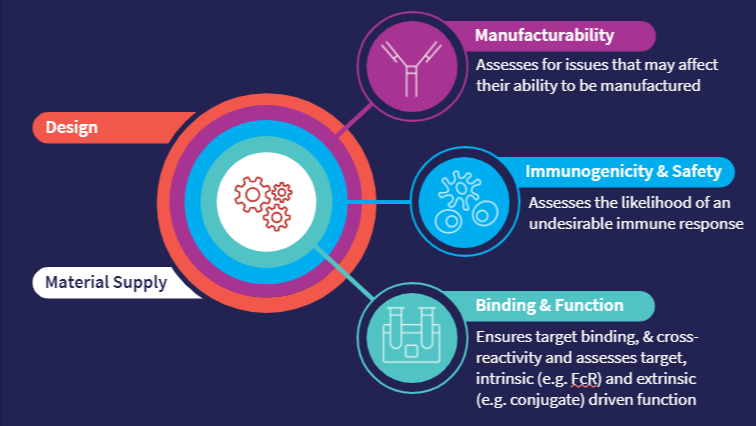
By asking, “Can we make it?” and “Does it work?” right from the start, we can begin to whittle down candidates from hundreds to a handful. From there, we can keep refining the selection criteria, continually derisking a project and moving closer to that ideal drug.
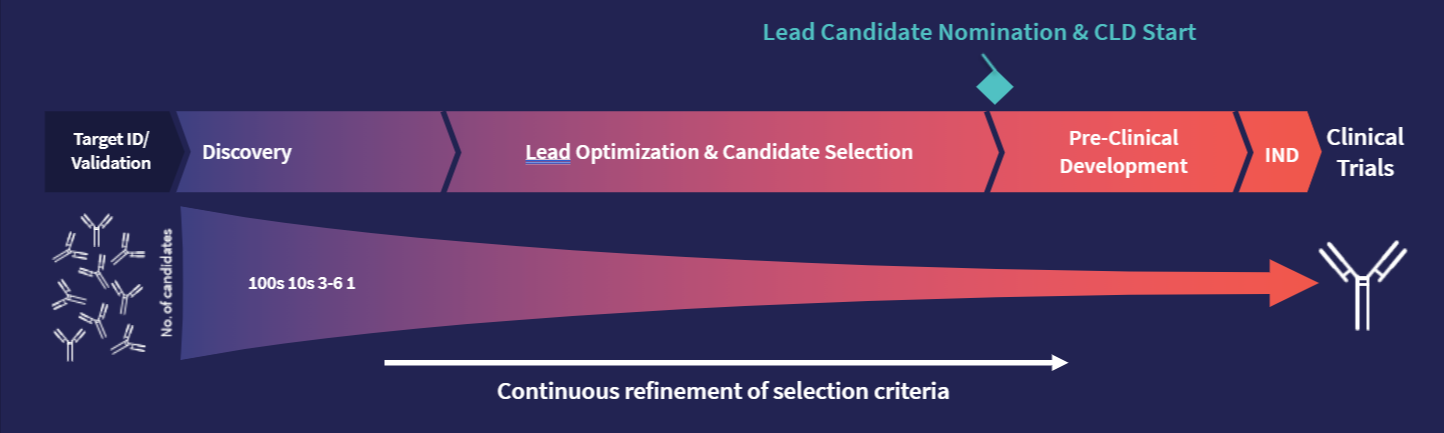
Hit generation followed by high throughput in silico and in vitro screening to screen out candidates with undesirable properties.
Candidates are designed and engineered to remove undesirable properties and introduce desirable properties.
Detailed biological and biophysical characterization of a small panel of molecules in order to select a lead candidate for progression.
Comprehensive understanding of a single candidate and capability to develop, manufacture and deliver your drug for both clinical and commercial scale.
Developability Assessments are critical to enabling effective lead candidate selection. In summary Developability offers:
Now you know more about what goes into the developability process at Abzena, what exactly can you get? Well, we offer both flexible pre-defined packages, or tailored à la carte options so you can get exactly what your project needs and address specific questions.
Are you ready to derisk and develop your next biologic or bioconjugate with us? Get in touch and let’s work together to move medicine forward faster.
Developability refers to the likelihood that a biologic or bioconjugate can become a manufacturable, safe, and efficacious drug. Developability assessment measures those characteristics of a drug candidate that make it suitable for development and manufacturing at scale – helping reduce project risk and increases the chances of advancing a molecule to regulatory approval.
Bringing a biopharmaceutical from discovery to market is challenging and costly. Without early developability assessment, many promising molecules fail due to manufacturability, safety, or efficacy issues. Assessing early helps de‑risk development, manage timelines and budgets, and improve success rates.
Abzena’s approach centers on two fundamental questions:
These questions guide early molecule triage and optimization.
Abzena blends decades of complex biologics design experience with stage‑appropriate assays to refine candidate selection criteria over time. This ensures large molecules are continually evaluated and optimized as a project progresses.
Abzena’s developability platform solution centers on four key scientific areas:
Abzena uses a suite of predictive and empirical tools, including:
These tools help predict and mitigate potential immune responses early.
Manufacturability is assessed by:
Abzena provides stage‑appropriate packages, including:
Abzena’s design and optimization capabilities for complex biologics span:
Bioconjugates including Antibody-Drug Conjugates (ADCs), Antibody-Oligonucleotide Conjugates (AOCs) and Radionuclide Drug Conjugates (RDCs)