Why Abzena?
Trust our focused approach.

Move forward to the clinic or market faster with our specialized, integrated expertise in complex chemistry and advanced bioconjugation services.
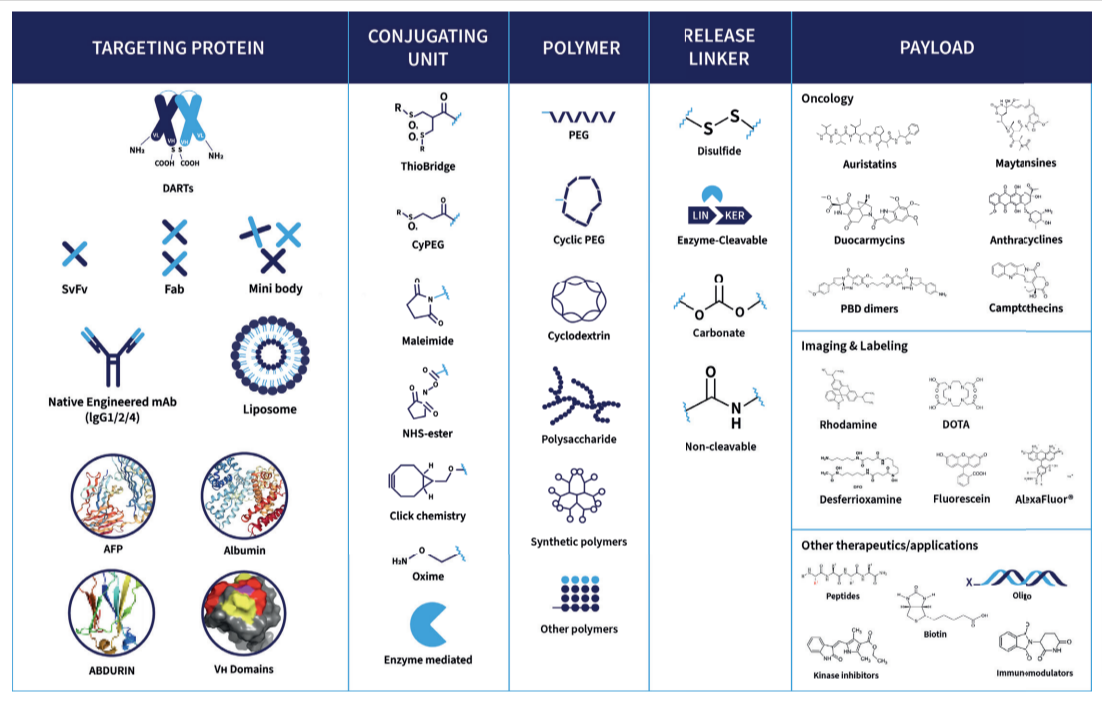
Bioconjugates such as ADCs, Bispecific ADCs, Dual-Payload ADCs, AOCs, and RDCs, are powerful modalities with complex development needs.
Abzena is at the forefront of next‑generation bioconjugate innovation – including dual‑payload strategies designed to enhance efficacy and overcome drug resistance. With fully integrated teams and streamlined end-to-end solutions, we simplify complexity, accelerate development, and move your program efficiently toward clinical and commercial success. We specialize in:
We ensure a robust and consistent bioconjugate manufacturing process from clinical to commercial scale. Our capabilities include:

“Abzena has deep expertise in the design of ADC linkers. We leverage our bioconjugation toolbox to optimize ADC stability, hydrophilicity, drug loading, and the drug-release mechanism. Abzena has access to a large range of cytotoxic payloads. We have expertise in developing ADCs with novel payloads for applications in both oncology and other therapeutic areas. Abzena provides bioconjugation and linker solutions to create dual-payload ADCs.”
Linker technologies are critical when developing Antibody-Drug Conjugates (ADCs). Most linkers, particularly those used in first-generation ADC bioconjugation have limitations. The impact, safety and efficacy of the ADC can be compromised. ThioBridge® was developed by Abzena to overcome those limitations and improve the design and delivery of ADCs.
Building on the unmatched expertise of our scientists, the IP we’ve generated, and our advanced facilities in the UK and US, we have everything you need to implement fully integrated commercial bioconjugation & ADC development and manufacturing programs.
We offer access to genuine scientific experts, streamlined development solutions, interconnected teams and full end-to-end support – all under one organization.

Bioconjugation has emerged as a cornerstone technology, enabling precise drug delivery, whilst enhancing therapeutic efficacy and reducing adverse effects. In our latest whitepaper our bioconjugation experts have explored the variety of conjugate types that are now accessible, each designed to enhance medical outcomes in various therapeutic areas.
Moving medicines forward created with bioconjugates such as ADCs, AOCS and RDCs, and linker payloads is where we excel. Abzena is the leading, fully integrated, bioconjugate CDMO + CRO services provider through to commercialization.

At every development stage, Abzena offer stage-appropriate integrated solutions for bioconjugates that deliver on your immediate development goals. Our focus is always on finding the right solution, at the right development stage whilst reducing risk and ensuring future scalability downstream.
Our stage appropriate solutions are designed to help your program move from Hit-to-Lead candidate discovery faster or accelerate DNA-to-IND to CTA enabling programs and beyond to de-risking commercial launch.
Bioconjugates are hybrid molecules linking biological and chemical components. They include antibody-drug conjugates (ADCs) and antibody-oligonucleotide conjugates that can be used in highly targeted therapies.
Bioconjugation or bioconjugate chemistry as it can also be referred to, is the chemical process of precisely linking two (or more) molecules, where at least one component is a biologically active molecule with a potent payload. The goal of bioconjugation is to create a new and stable targeted therapy, a ‘bioconjugate’ such as an ADC or AOC.
Yes, Abzena has experience through multiple AOC programs, with expertise in antibody formats (mAbs, Fabs, VHHs), analytics and biological assessment such as cell binding, trafficking and gene knock-down.
ADCs and AOCs share structural similarities. They both combine antibodies with functional payloads. Their development processes differ significantly due to the complexity of oligo payloads in AOCs. These differences present distinct technical, analytical and regulatory challenges that experienced developers like Abzena must address.
The process starts by defining the molecule’s function and development stage, then designing tailored conjugate panels to guide discovery and optimization.
For AOCs, regulatory frameworks are continuing to evolve. Developing AOCs involves navigating a regulatory landscape that is incomplete. The FDA and EMA classify oligos differently. The unique characteristics of AOCs – particularly their oligo payloads – present challenges that existing frameworks do not fully address. This gap requires experienced bioconjugate developers such as Abzena to adopt proactive and scientifically rigorous approaches.
Yes, if a suitable handle is available for linker attachment, Abzena can conjugate your payload to a monoclonal antibody and guide you through the process.
Yes, Abzena offers end-to-end tech-transfer support for ADC, AOC, and other conjugation programs, helping companies onshore their processes while maintaining consistency, quality, and scalability.
The leading biologics CDMO + CRO that accelerates drug development, moving medicine forward for patients in need.