Why Abzena?
Trust our focused approach.
Authors: Dr. Rob Holgate, Vice President of Research & Innovation, and Dr. Gary Watts, Director of Formulation Development at Abzena
In Part 1, we looked at the importance of developability assessments and the role they can play in providing you with a greater understanding of your drug candidates before committing to preclinical development. Moving beyond initial discovery and screening, the next critical phase is lead optimization – an iterative process where candidate molecules are refined to balance potency, safety, and manufacturability.
In Part 2, our experts, Dr. Rob Holgate, Vice President of Research and Innovation, and Dr. Gary Watts, Director of Formulation Development, explore how design and developability go hand in hand, how data drives rational design, the scientific strategies and tools used, and how multidisciplinary teams make informed decisions that shape the future of next-generation biologics.
The journey from a pool of initial hits to a single clinical candidate is one of refinement and elimination. However, at Abzena, before performing any developability assessment, we establish a solid foundation through good design.
Candidate design takes many forms and depends on what you’re starting with and what your end goal is. As mentioned in Part 1, having a TPP in place helps frame how you want the molecule to look, which in turn guides the required design effort.
Examples of common design considerations are outlined below:
As can be seen, design can take many forms, and often, multiple design concepts will need to be addressed into a single molecule. However, each round of engineering – whether for improved affinity, stability, or reduced immunogenicity – will likely change the biophysical and functional profile of the molecule. Therefore, developability shouldn’t be something you perform as an afterthought on the final couple of lead candidates; it should be viewed as a continuously applied process that complements the design process.
The best molecule is not always the one with the highest in vitro potency; rather, it is the candidate with the optimum combination of biological activity, safety, and manufacturability.
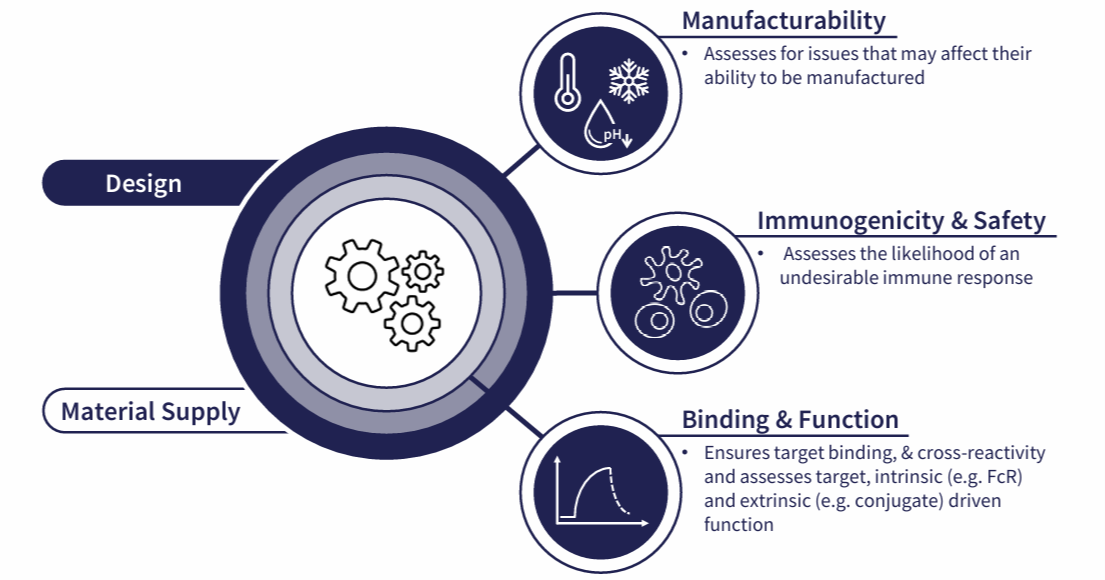
To achieve this, we focus on three aspects:
Fig 1. Candidate selection requires a thorough understanding of your candidate molecule
Before we move on to talk about the different assessments, we should highlight that, irrespective of the assessment, being able to generate appropriate quantities of high-quality material in an appropriate timescale is key to generating reliable, reproducible results.
The most common approach is to use material from transients at the early stages, since this tends to be quicker and cheaper. Commonly used systems include platforms such as the ExpiCHO transient expression to generate low mg to 500+ mg quantities.
Typically, though, these platforms are surrogates for the final manufacturing cell line and so, getting into the final manufacturing system as early as possible is increasingly becoming the trend when it comes to derisking. To this end, bringing stable pools based on the manufacturing cell line, for example, our AbZelectPRO CHO-K1 cell line, earlier into development is becoming increasingly common. Whilst not the final clone, the material is more representative than earlier transient material.
Fig 2. Providing a thorough understanding of your candidate molecule
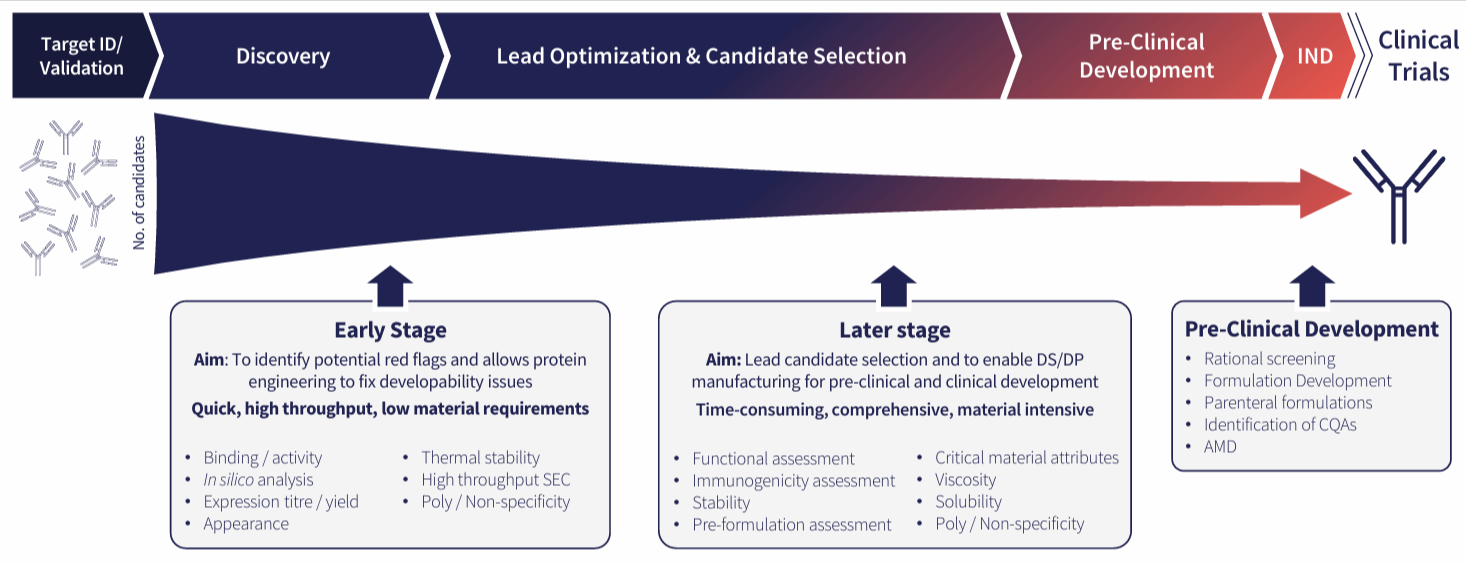
At earlier stages of drug discovery, typically you will have many potential candidates (10s to 100s) but only very low amounts of each (0.1-0.5 mg), so you really want to maximise the information you can get with what limited material you have. One of the main aims at this stage is to get rid of large numbers of unsuitable candidates as quickly as possible to allow a more detailed deep dive on a smaller number of potential candidates. The assessments outlined below are focused primarily on biophysical attributes rather than function since functional assays are likely to be specific for particular targets, whereas the assays described below are agnostic of target.
A useful approach at this stage is to use in silico tools since they can provide rapid and high throughput assessments, allowing you to rank and prioritize variants as necessary based on sequence. Two of the main ways in which in silico tools can be used at this stage include assessing potential manufacturability and potential immunogenicity of sequences.
Manufacturability: A number of in silico tools focus on analysing the primary sequence of candidates for potential sequence liabilities (e.g., deamidation or isomerization sites) or determining phys-chem properties such as pI. Next-level tools can be used to build in silico structural models that can be used to identify problematic charged or hydrophobic regions that have the potential to impact biophysical properties. Other tools can also be applied, e.g., Therapeutic Antibody Profiler (TAP) allows you to determine how your molecules stack up against a database of clinical-stage therapeutics by assessing a number of parameters that are thought to be implicated in poor developability.
Immunogenicity: T cell epitopes are specific peptide fragments that are recognized by T cell receptors when presented by MHC class II receptors. These can lead to an immune response and, in the context of therapeutic proteins, the development of anti-drug antibodies (ADAs). In silico tools, such as Abzena’s iTope-AI, are used to identify potential T cell epitopes present within a sequence, and can be used to help rank candidates, and identify potential hotspots that may be of immunogenic concern, allowing re-engineering if needed.
It should be noted that in most cases, in silico tools are inherently over-predictive but nonetheless remain invaluable when it comes to ranking variants as well as assessing the impact of any potential design changes before a molecule is even made. It remains, however, that actual wet work provides the real-world experimental data that really drives downstream decision making.
It is sometimes not fully appreciated, but data gathering starts from the very initial expression and purification and can start to provide insights into a molecule’s manufacturability. For example, low titres and/or QC data showing high levels of aggregation (e.g., as determined by analytical SEC) using a high-expressing system such as ExpiCHO can provide a red flag.
Following on from expression, there are multiple ways of using this purified material effectively to help narrow down candidates. Some of the more common methods are outlined below, each looking at a different attribute of the candidates.
Thermal ramp stability measurements (typically Tm and Tagg) are well-established methods for looking at the stability of proteins in, e.g., different formulations. However, at early stages of development, the readouts are mainly used to rank variants. Tm indicates the point where the protein unfolds, while Tagg identifies the temperature at which the protein begins to aggregate. Combining both readouts provides valuable insights into molecule stability and propensity for aggregation. Importantly, the material requirement is very low, with information available using as little as ~10 μg of sample, throughput is reasonable (up to 48 samples at a time), and the time to get data is relatively short (<1 day).
By contrast, techniques such as HIC (hydrophobic interaction chromatography) and AC-SINS (affinity-capture self-interaction nanoparticle spectroscopy) focus on assessing different molecular aspects such as aggregation and self-association propensity. Techniques such as HIC are used for early-stage assessments, where the focus is to look at the relative hydrophobicity of molecules, often against a panel of clinically approved mAbs. Red flags would be candidates that fall outside of the expected range – particularly hydrophobic molecules are more likely to have manufacturing challenges associated with, e.g., aggregation or viscosity. AC-SINS is a method for determining self-interaction, a useful parameter to measure since high self-interaction can lead to high viscosity and low solubility, which can impact certain formulations and routes of administration. Again, these have low material requirements and relatively good throughput, allowing many candidates to be screened in a short timeframe.
Fig 3. Highlighting an example of a HIC experiment and a thermal ramping experiment
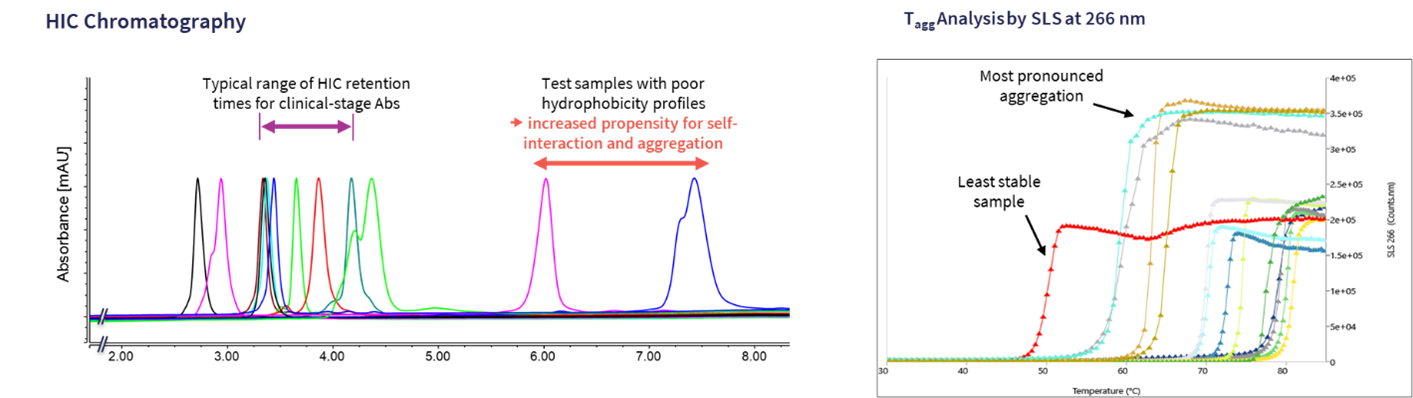
The left panel shows a series of antibodies and their absorbance normalized HIC retention time. The majority show retention times within the range exhibited by clinical stage antibodies or approved antibodies run within the same experiment, suggesting an acceptable level of hydrophobicity. There are, however, a couple of samples on the right-hand side that show significantly higher hydrophobicity, which would be of concern if not addressed
The right panel shows an example of a thermal ramping experiment using static light scattering as the readout for Tagg determination. We can observe a wide variety of aggregation propensities profiles from the sample with the lowest Tagg (red profile) to more stable samples towards the right-hand side of the graph. The magnitude of the response also indicates the degree of aggregation.
Whilst not looking at manufacturability directly, Polyreactivity is an indirect measure of biophysical properties and refers to the ability of antibodies to bind multiple unrelated antigens – essentially a measure of how “sticky” a molecule is. This can pose risks for PK and safety, so assessing this early on is essential. Tools such as ELISAs against baculoviral particles or a panel of unrelated antigens such as insulin, dsDNA, etc., can provide a rapid, early assessment of the likelihood of polyreactivity.
What you do with this information from these assessments will vary, most likely depending on, for example, how many candidates you have. If you are fortunate to have a diverse panel of candidates, these assessments may allow you to triage down and discard candidates with a poor profile and concentrate on a small set of variants. If, however, you have a limited number of candidates, this information can help prioritize which ones you need to focus efforts on.
If any issues are highlighted, then a typical approach would be to remove these potential issues using a range of different protein engineering techniques that are available. As discussed in Part 1, it’s important to highlight that while it may seem costly and time-consuming to perform reengineering at this stage, it is far more cost-effective than trying to then re-engineer a drug that has already gone through Phase I, where the price of failure is significantly higher.
Sometimes, antibody engineering may not be possible. It could be that a site is a critical motif for potency, and it can’t be changed. Or it could be for other reasons, such as intellectual property, where a proposed sequence change may clash with someone else’s IP. If that is the case, other approaches may be required. In this situation, one common approach is to explore whether formulation development can be a viable option to overcome the issue observed.
Effective lead optimization in biologics development is impossible without continuous, data-driven developability assessments linked with good design. Integrating knowledge and a range of refined approaches ensures that only the most robust, manufacturable, and safe candidates advance. By balancing function, immunogenicity, and manufacturability and proactively addressing issues, teams can reduce broader risks for Preclinical and clinical development.
In the final installment of this series, we will examine how later-stage developability assessments support candidate selection, clinical and commercial readiness, focusing on advanced analytical testing, manufacturability at scale, and lessons learned from case studies.
Missed Part 1 of this series? Discover ‘The Importance of Developability Assessments – What is the critical role of developability assessments for guiding the next generation of drugs?’