Why Abzena?
Trust our focused approach.
Authors: Dr. Rob Holgate, Vice President of Research & Innovation, and Dr. Gary Watts, Director of Formulation Development at Abzena
The path from early discovery to a successful biologic drug is fraught with scientific, technical, and commercial challenges. Despite revolutionary advances in antibody design & engineering, synthetic biology, and analytical characterization techniques, most biologics never reach the market. The primary reasons for this high attrition rate include not only issues with efficacy or toxicity, but also critically overlooked risks in manufacturability, stability, and immunogenicity. Ignoring these key areas in the early stages of drug development can leave drug developers vulnerable with a less-than-ideal candidate to progress onward toward the clinic.
In this three-part blog series, Abzena’s Dr. Rob Holgate, Vice President of Research and Innovation, and Dr. Gary Watts, Director of Formulation Development, look at how developability assessments can provide you with a greater understanding of your drug candidates before committing to preclinical development. Learn how developability assessments can save time and money by allowing scope for early risk mitigation or corrective engineering. We’ll also look at how these assessments can offer the highest chance of progressing your successful lead candidate to the clinic.
In the first part of our three-part series, we explore why developability assessments are a critical – yet sometimes underappreciated – component of the candidate selection process in biologics development. While efficacy and target engagement remain important, this post will highlight how early assessment of manufacturability, safety, and risk can set the stage for more successful, efficient drug development. We’ll introduce the concept of developability, common reasons for candidate attrition, and how early risk identification is key to avoiding costly late-stage failures.
At Abzena, we understand that drug development is a complex endeavor with multiple stakeholders. On the one hand, we are trying to develop the “perfect” drug, i.e. a molecule that has all the right properties within a single entity – high drug stability, highly efficacious and with no undesired safety or toxicity challenges. However, balanced against this are the realities of drug development – constant pressures on timelines and getting a drug into the clinic as soon as possible, coupled with the high financial cost of developing increasingly complex molecules.
The unfortunate reality is that many of these drugs will fail. Industry data suggests that only about 10% of biologic drug candidates that enter clinical development ever gain approval. Most failures occur between discovery and Phase II, often at the point where proof of concept (POC) is established.
The reasons for failure can be multiple, however principal amongst these include a lack of clinical efficacy or unmanageable toxicity, potential safety issues, or immunogenicity issues. Alternatively, or in addition, some may show poor drug-like properties in terms of being able to make them at scale and to the required quality. Then, outside of the scope of this article, there are more business-driven reasons in terms of lack of commercial need, or poor strategic planning.
Irrespective, the impact of failure is high! Development delays and failures normally lead to unplanned costs associated with redesign and testing. This in turn puts strains on finances and can damage investor confidence and a company’s ability to secure future funding. Ultimately, however, the biggest impact is in the ability to deliver life-changing medicines.
At Abzena, what we want to do is reduce this attrition. Abzena’s mission is to move medicine forward, and so our objective is to increase the chance of your molecule successfully making it through clinical trials. Any attrition is going to delay the delivery of these life-changing medicines to patients in need.
Traditionally, candidate selection has prioritized measures such as binding affinity, functional potency, and in vivo efficacy. These attributes are, of course, essential. However, experience has shown that candidates with promising activity profiles can fail later in development due to poor biophysical properties, manufacturing challenges, or immune responses in patients. This is where Developability comes in.
Developability is a multi-dimensional concept encompassing the technical feasibility of advancing a biological candidate from the laboratory to the clinic and, ultimately, the market.
Developability is essentially trying to answer two simple questions. We are looking to understand, can we make it, and does it work?
1. Can we make the molecule?
Firstly, can we make it in the lab, where we’re typically looking at it at small (typically mg) scale. Following this, the object is to de-risk the process as we scale up through CMC production where we’re dealing with a completely different scale of production, often kilograms of material from thousands of liters of culture media. We are trying to de-risk this transition from the small-scale lab through to the CMC process.
2. Does it work?
We need to ensure that the molecule has the desired mechanism of action and that if we change anything, whether through e.g. deliberate engineering or through aspects such as changing formulation we’ve not impacted this. Furthermore, we need to ensure the molecule has an acceptable PK, safety and immunogenicity profile, and more, which we discuss in Part II of this series.
As we apply developability, it becomes apparent that identifying the best candidate requires a holistic approach, investigating multiple parameters and aspects of the molecule.
Assessing and optimizing all of these attributes is critical for clinical and commercial success. Ignoring them can result in costly failures, even for candidates with strong activity profiles.
Developability isn’t a static process. Rather, it is a continuum – it starts all the way at the beginning when you’re selecting your initial discovery candidates of interest through to the point at which you nominate your lead candidate to move into preclinical development, and sometimes beyond.
With this in mind, selecting the most appropriate assay for a particular stage of development is key.
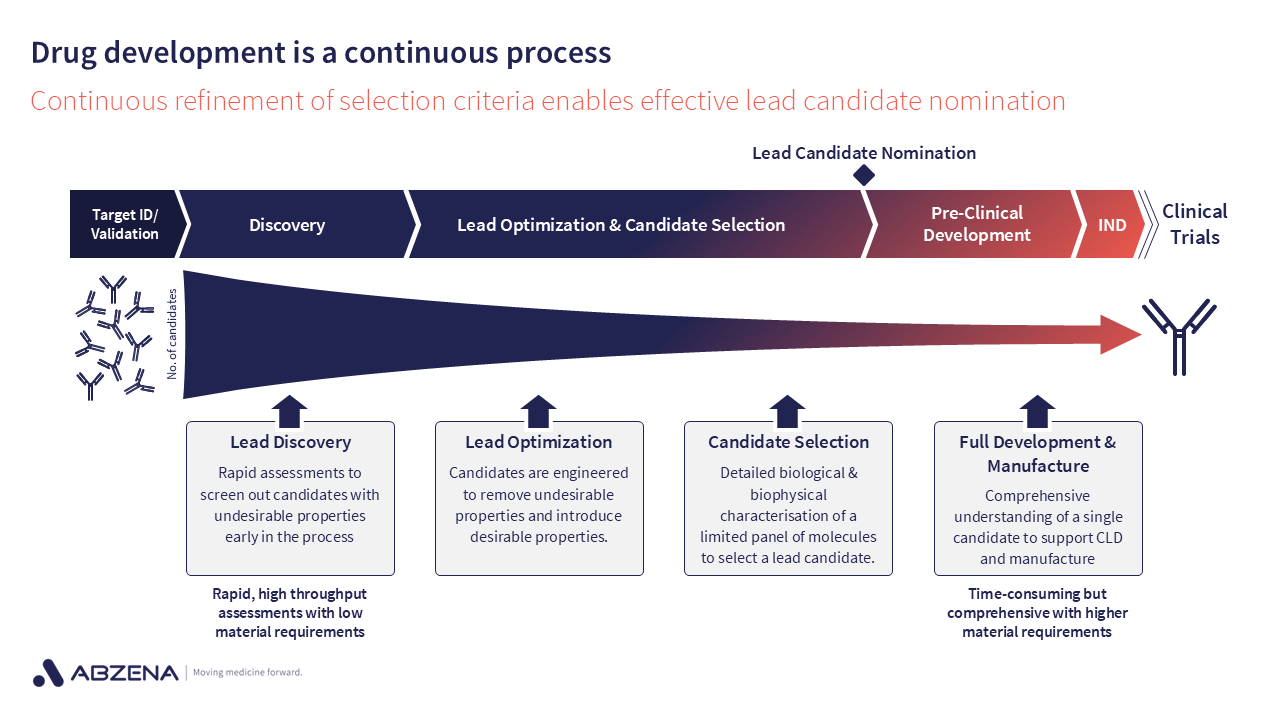
Over the last several years an approach has evolved to incorporate developability screening at earlier stages of the Discovery & development pathway, reflecting a broader shift in the industry towards bringing risk visibility earlier.
The “fail fast, fail early” philosophy has become a guiding principle in drug development. The earlier a risk is identified, the less costly it is to address – whether that means engineering a better molecule, adjusting formulations, or even discontinuing a program before major investments are made – failing fast, failing early, is going to be far better than failing in phase II or phase III, where the cost of failure is that much higher. In this context, performing assessments help organizations allocate resources efficiently, reduce financial risk, and provide confidence to investors or partners.
Robust developability assessments are not just about identifying risks; they are also about providing solutions. In this regard, developability and design are intricately linked!
Derisking options may include re-design and re-engineering of the molecule to remove the issue in question. However, when risks cannot be eliminated through engineering, they must be acknowledged and managed—sometimes by adjusting processes, e.g. choosing a more suitable formulation or, in some cases, through careful monitoring in later development.
Common liabilities and mitigation strategies include:
Abzena has the expertise to provide robust developability assessments and solutions that help address these risks. We have the capabilities to provide a thorough understanding of your molecule, identifying risk factors as early as possible in the development process. As we identify these risk factors, we provide the scope to be able to fix these or reduce them where possible. If we cannot fix them, at least we have that knowledge; we know that they are there, and we can try to manage these liabilities.
At the beginning of any program, it is essential that you always consider the end goal – what does the final drug candidate need to look like?
One useful approach is to use strategic tools such as establishing a Target Product Profile (TPP) or Candidate Drug Target Profile (CDTP). A TPP serves as a blueprint for the entire development plan, outlining intended use, dosing, route of administration, and target patient population. A CDTP is similar, but is focused primarily on the R&D phase, providing clear specifications for what the ideal molecule should look like at each stage.
These documents can help shape the assessments that are needed as molecules progress. For example, do you need a high concentration formulation? If so, this should be factored into the manufacturability assessments as early as practicable to avoid progressing too far to then finding out this is not achievable.
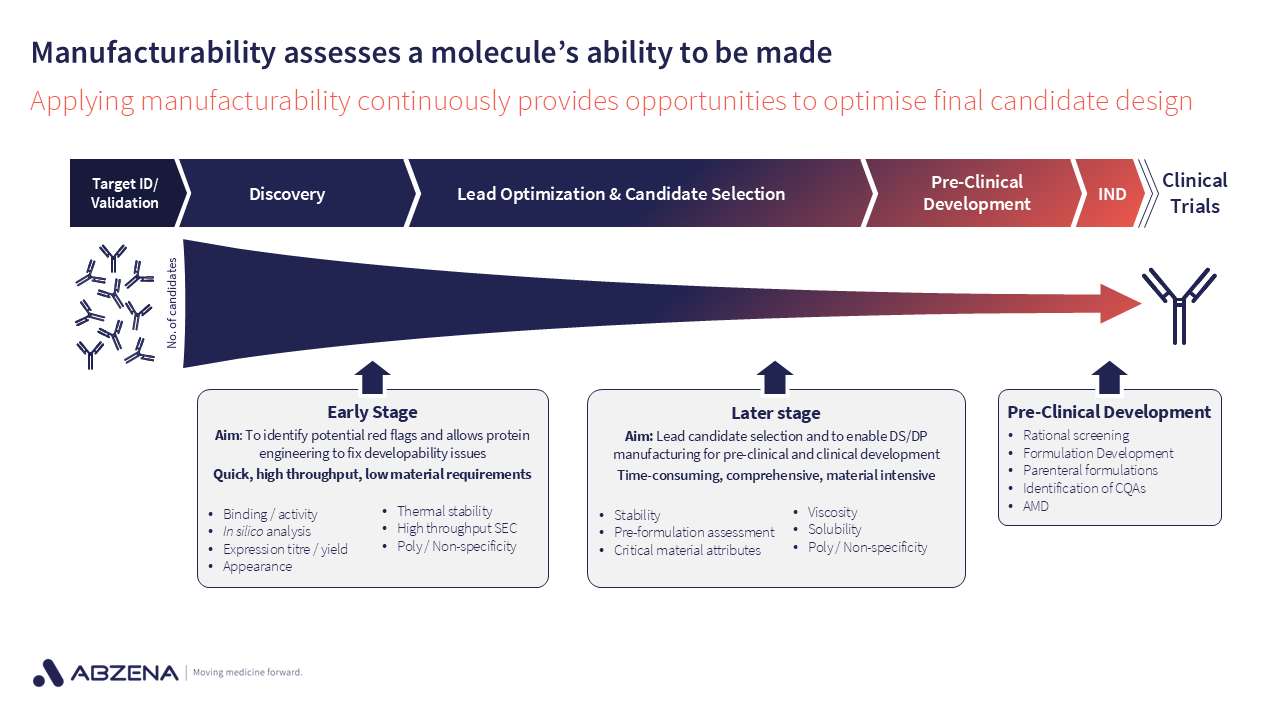
As new data emerges, it helps teams stay aligned and make data-driven decisions on when to advance, optimize, or deprioritize candidates.
This table some of the aspects that we look at as part of CDTP or TPP.
Indication
| Properties | Details |
| Patient Population |
|
| Modality |
|
| Dosage & Administration |
|
| Commercial Strategy / Approach |
|
| Pharmacological Properties & Efficacy |
|
| PK & PD |
|
| Safety |
|
An optimal biologics development workflow integrates developability assessments from the earliest stages:
Cross-functional collaboration is key – protein engineers, analytical scientists, manufacturing experts, clinical teams, and regulatory specialists and many others must all contribute to the developability roadmap.
As antibody pipelines continue to expand and discovery technologies evolve, developability assessments are no longer a luxury. They have become a necessity for successful biologics development.
By understanding their critical role and integrating these assessments from the outset, organizations can:
In the next part of our series, we will explore how developability assessments guide molecular design and lead optimization, providing a deeper dive into the strategies and technologies that enable robust candidate selection.