Why Abzena?
Trust our focused approach.
From payload optimization to integrated GMP manufacturing, companies aim to elevate oncology ADCs to standard of care.
Antibody-drug conjugates (ADCs) constitute a rapidly evolving class of targeted oncology therapeutics that couple tumor-selective antibodies with highly potent cytotoxins. At the 16th Annual World ADC Conference, leaders across discovery, development, and manufacturing highlighted the accelerating momentum of this clinically maturing field.
Presentations focused, for example, on expanding payload diversity, advancing site-specific conjugation, and engineering hydrophilic linkers to improve stability. Others addressed implementing scalable, stage-agnostic Chemistry, Manufacturing, and Controls (CMC) strategies and integrating manufacturing models for increasingly complex bioconjugates. In this landscape, contract research organizations (CROs) and contract development and manufacturing organizations (CDMOs) play an increasingly central role in advancing ADC candidates from concept to clinic.
In the quest to produce the optimally engineered ADC, a key complication is that conventional conjugation chemistries attach the cytotoxic payloads randomly on an antibody, producing a mixture of functionally variable heterogeneous ADC molecules.

Nicolas Camper, PhD, vice president of ADCs and bioconjugates for CRO/CDMO Abzena, says the company offers a new platform to help solve such challenges. “Inadequate choices of bioconjugation and linker technologies can derail an ADC program. Using a bioconjugation and linker platform such as Abzena’s ThioBridge™ site-specific conjugation technology ensures that the ADC generated has favorable pharmacokinetics, biodistribution, and manufacturability properties, independent of the selected antibody and payload, giving the ADC the best chances of success in the clinic.”
The process involves selectively reducing antibody interchain disulfide bonds and then treating them with bis-sulfone reagents that covalently rebridge the reduced disulfides. Camper explains, “This allows for the site-specific conjugation of linker-payloads at antibody interchain disulfides to generate ADCs with precise, homogenous drug loading. ThioBridge ADCs are very stable with no deconjugation of the linker-payload observed during storage or in systemic circulation.”
The linkers also incorporate a hydrophilic polymer in their structure. Camper notes, “This enhances the biophysical stability of the ADCs and decreases their propensity to aggregate. Importantly, this also reduces ADC hydrophobicity and imparts favorable pharmacokinetics to the conjugates.”
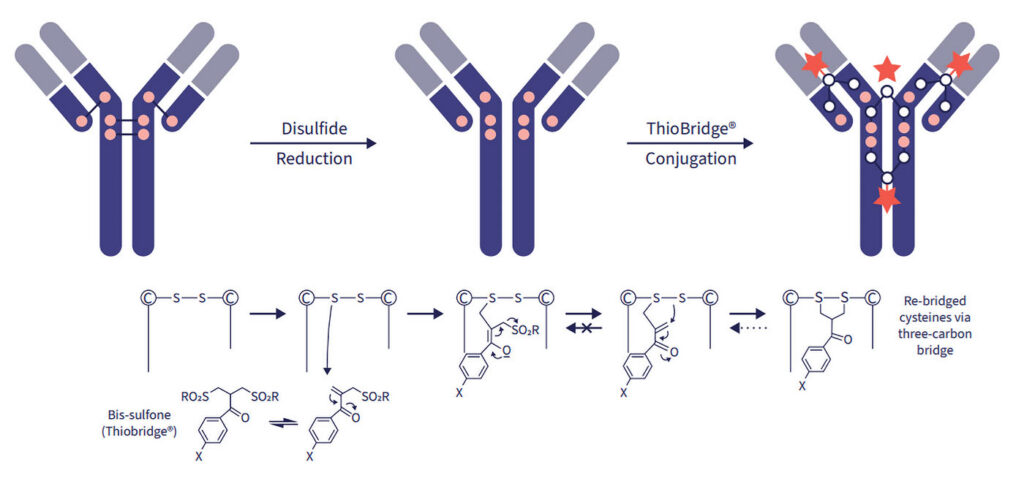
To more precisely link cytotoxic payloads to antibodies, Abzena’s ThioBridge™ platform selectively reduces interchain antibody disulfide bonds and treats them with bis-sulfone reagents to create site-specific conjugation of linker payloads for ADC.
As to the ease of manufacturing of these ADCs, Camper says an advantage of the platform is that it can prepare ADCs from native antibodies. “These are much more straightforward and far less costly to produce than engineered antibodies that, for instance, may incorporate engineered cysteines or non-natural amino acids. Overall, ThioBridge is a conjugation technology well-suited to the low-cost manufacturing of ADCs and is well validated. We now have three customers evaluating ThioBridge ADCs in clinical trials, and we continue to develop linker formats to accommodate the increasing diversity of payloads.”