Why Abzena?
Trust our focused approach.
Author: Dr Unmesh Lal, VP of Healthcare & Life Sciences at Frost & Sullivan
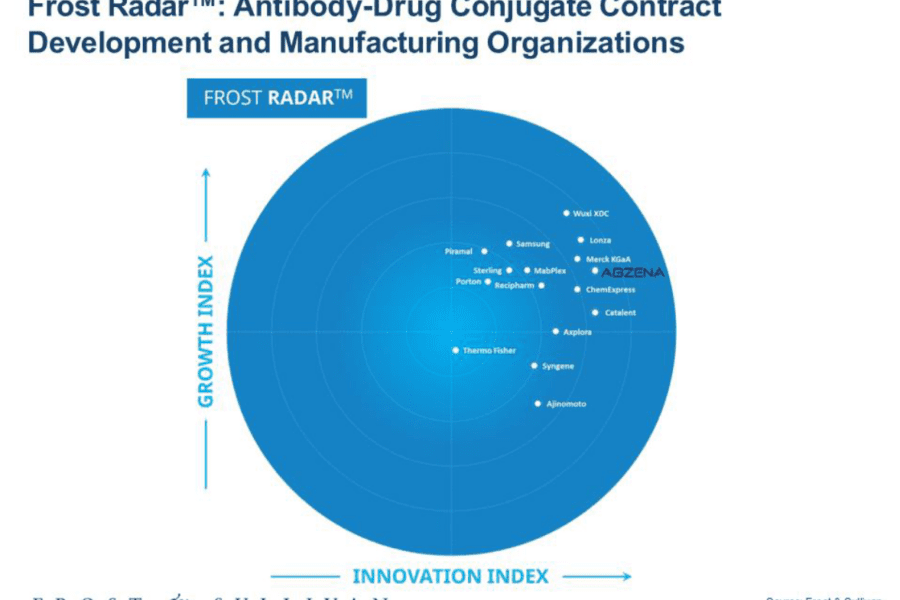
The global antibody‑drug conjugate (ADC) landscape is undergoing rapid expansion, fuelled by a surge in clinical pipeline progression, next-generation payload innovation, and the intensifying need for integrated development and manufacturing partners. Within this competitive environment, Abzena has secured a prominent position in Frost & Sullivan’s Frost Radar™: ADC Contract Development and Manufacturing Organizations, 2025, reflecting the company’s scientific depth, specialized capabilities, and differentiated technology platforms.
Frost & Sullivan’s 2025 assessment identifies a clear shift in CDMO selection criteria: sponsors increasingly prioritize partners capable of delivering specialized bioconjugation chemistry, biologics development, high‑potency handling, and advanced analytics within a single integrated framework. Abzena demonstrates strength across each of these dimensions.
Its dual‑continent operating model, San Diego (biologics production), Bristol, PA (HPAPI & conjugation), and Cambridge, UK (R&D and analytical platforms), offers global sponsors a harmonized route from discovery to early clinical and commercial‑ready manufacturing. This footprint enhances regulatory alignment with both FDA and EMA requirements and mitigates geographic supply‑chain risk.
Abzena’s proprietary ThioBridge® conjugation technology is highlighted in Frost & Sullivan’s Radar as a defining competitive differentiator. The platform enables stable, site‑specific, and homogeneous ADCs with precise DAR control, a requirement for improving therapeutic index, manufacturing reproducibility, and regulatory predictability.
The company further strengthens early‑stage workflows through AbZelectPRO™, an accelerated, high‑performance cell‑line development engine. Advanced analytics including EpiScreen® 2.0 for immunogenicity prediction and LabZient™ for structural characterization provide deep functional insights into increasingly complex conjugate architectures.
These capabilities collectively position Abzena to support the full spectrum of emerging modalities highlighted in the ADC CDMO Frost Radar, including ADCs, AOCs, RDCs/RACs, and next-generation hybrid constructs.
Frost & Sullivan notes Abzena’s strong record of program continuity, with many partners advancing from discovery through Phase I and beyond within the same operational network. This “grow‑with‑the-client” pattern underscores high satisfaction, operational reliability, and strong technical governance, reducing handoff‑related delays that frequently affect multi‑vendor outsourcing models.
The integrated execution model is particularly valuable for small and emerging biopharma companies, which often require scientific co‑development, CMC guidance, and streamlined project oversight to accelerate IND timelines.
The Frost Radar highlights recent expansions at Abzena’s San Diego site, which added late‑stage and commercial‑ready biologics capacity, including large-scale single-use bioreactors. Meanwhile, Abzena’s Bristol, PA facility continues to serve as a central hub for high‑potency payload handling, conjugation, and final ADC manufacture, all under strict containment and quality systems.
These capabilities provide a differentiated, end-to-end continuum across:
This integrated network also supports the shift toward more potent payloads, diverse linker chemistries, and novel conjugate modalities identified in Frost & Sullivan’s assessment.
Globally, the ADC CDMO ecosystem consists of two major archetypes:
According to Frost & Sullivan’s Radar, Abzena stands out as a mid‑sized specialist distinguished by proprietary technology platforms, deep analytical capabilities, and strong clinical‑stage execution. Its scale enables the flexibility, responsiveness, and collaborative engagement model that biotech’s rely on to advance complex, precision‑engineered ADC programs with confidence.
Abzena’s placement on the Frost Radar™ underscores its emergence as a precision‑driven, innovation‑centric CDMO built to support the next wave of bioconjugate therapies. With strong technological underpinnings, an integrated operating model, and an expanding global footprint, the company is well positioned to serve developers advancing ADCs, AOCs, and other high‑value targeted modalities.
As the ADC market continues to mature and as sponsors seek partners with both scientific depth and integrated execution, Abzena represents a compelling and strategically important player positioned for sustained leadership in the ecosystem.
About the Author:
Unmesh Lal has 20+ years of healthcare industry expertise, comprising of strategy consulting, primarily with global life sciences companies. His expertise lies in tracking game changing companies, innovative business models, and disruptive technologies while serving as a growth coach for clients in pharmaceutical contract services and precision health. He has authored thought leadership articles and presented them at leading industry events including J.P. Morgan, Bio-Asia, Bio-IT, Bioprocessing Summit, CPhI Worldwide, Biotech Pharma Summit, Medica Health IT, Biotech Outsourcing Strategies cmc, Precision Med Exhibition & Summit, Global Bio & Pharma Plaza, and others. His in-depth understanding of the precision health ecosystem enables him to guide companies in identifying growth opportunities across contract research, development, and manufacturing services. He holds a master’s degree in biomedical engineering from the University of Michigan-Ann Arbor.